What is the only non-metal to be found on the left side of the periodic table?
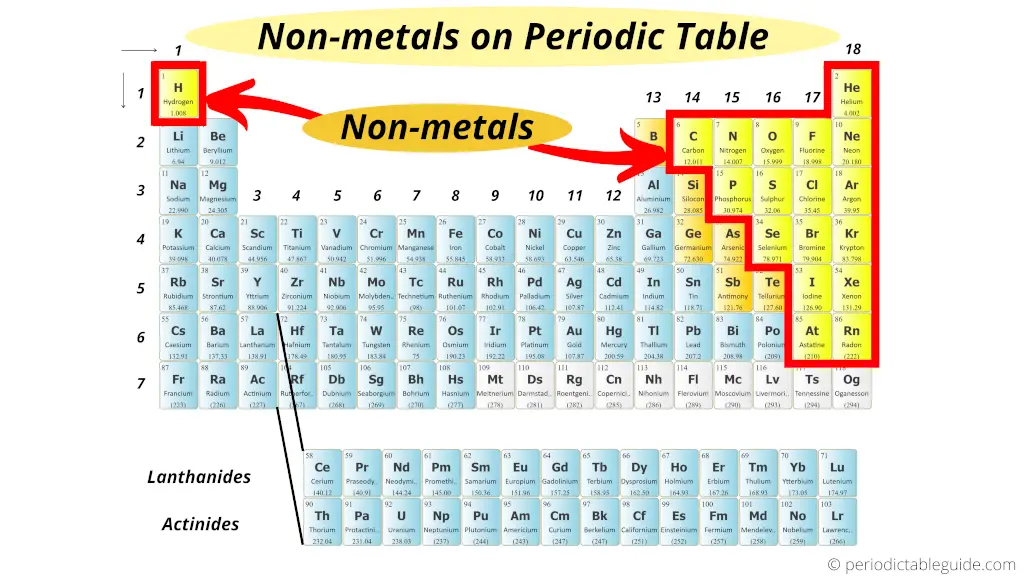
Nonmetals are located on the upper right side of the Periodic table (see above image).
Exception: Hydrogen is a nonmetal which is located on the left top corner of the Periodic tabular array.
Well, you take got the answer of "Where are Nonmetals located on the Periodic Tabular array?" But in that location are lot more than things that you should know almost the nonmetals like;
- What exactly are the nonmetals?
- How many nonmetals are there on the Periodic tabular array?
- List of nonmetals on Periodic tabular array
- Where are the most reactive nonmetals on the periodic table
- Properties of nonmetals on the periodic table
- Physical properties of nonmetals
- Chemical properties of nonmetals
Let us discuss all these topics apace.
Let's get started.
The simple answer: "Non metals" are "not" the "metals"
Or in other words nonmetals are those elements which do not possess the properties of metals.
Well, at present let me tell you lot the proper definition of nonmetals used in chemical science.
Nonmetals are the chemical elements which gain electron/southward during a chemical reaction and class a negative ion.
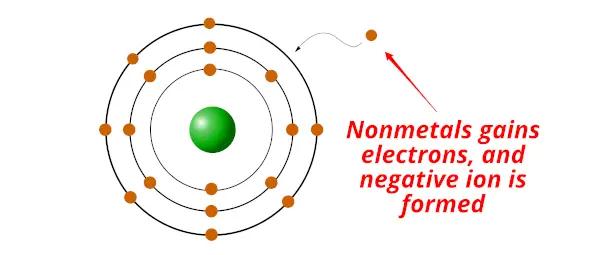
Non metals have exactly the opposite holding than that of the metallic.
In short call back that;
- Nonmetals are electrons gainers (nonmetals accept/gains electrons)
Elements which prove these properties are known every bit Nonmetals.
I accept discussed more physics and chemistry properties of nonmetals in this article only.
Stay tuned…
At present permit us see the total number of nonmetals on the Periodic tabular array.

There are 18 nonmetals on the Periodic tabular array.
All these nonmetals are located on the upper right corner of the Periodic table (Hydrogen is located on the left superlative corner)
In the above epitome, the nonmetals are represented in yellow colour.
[Note: Astatine (atomic number 85) shows characteristics of nonmetals (halogens) besides every bit metalloids. But many researchers accept found that it shows more similar backdrop like that of nonmetals and hence Astatine (At) is considered as a nonmetal (element of group vii) by many researchers] Source: HRW texts
And then on because Astatine as a nonmetal, at that place are total 18 nonmetals on the Periodic tabular array.
And if Astatine (At) is excluded from nonmetals, then at that place are full 17 nonmetals on the Periodic table.
But why is this number not exact?
Why is this number inexact?
The curt respond: Considering at that place is no widely agreed definition of metals, nonmetals and semimetals.
A pharmacist may ascertain nonmetals on the ground of chemic properties of the element, while a physicist may define nonmetals on the basis of density or atomic number.
So we can say that the total number of nonmetals on the Periodic table is 17 or eighteen.
State of nonmetals at room temperature
Nonmetals are found in solid land, liquid state besides as gaseous state.
- 11 non metals are in gaseous state at room temperature.
- Bromine (Br) is a nonmetal which is in liquid state at room temperature.
- Balance of the nonmetals are solids at room temperature.
Here is a complete list of all the xviii nonmetals on the Periodic table.
| Atomic number | Symbol | Proper name of element |
| 1 | H | Hydrogen |
| 2 | He | Helium |
| vi | C | Carbon |
| vii | North | Nitrogen |
| 8 | O | Oxygen |
| nine | F | Fluorine |
| 10 | Ne | Neon |
| 15 | P | Phosphorus |
| 16 | South | Sulfur |
| 17 | Cl | Chlorine |
| xviii | Ar | Argon |
| 34 | Se | Selenium |
| 35 | Br | Bromine |
| 36 | Kr | Krypton |
| 53 | I | Iodine |
| 54 | Xe | Xenon |
| 85 | At | Astatine |
| 86 | Rn | Radon |
Continue reading… more than important topics are on the style.
The short respond: Fluorine is a highly reactive nonmetal on the entire Periodic table.
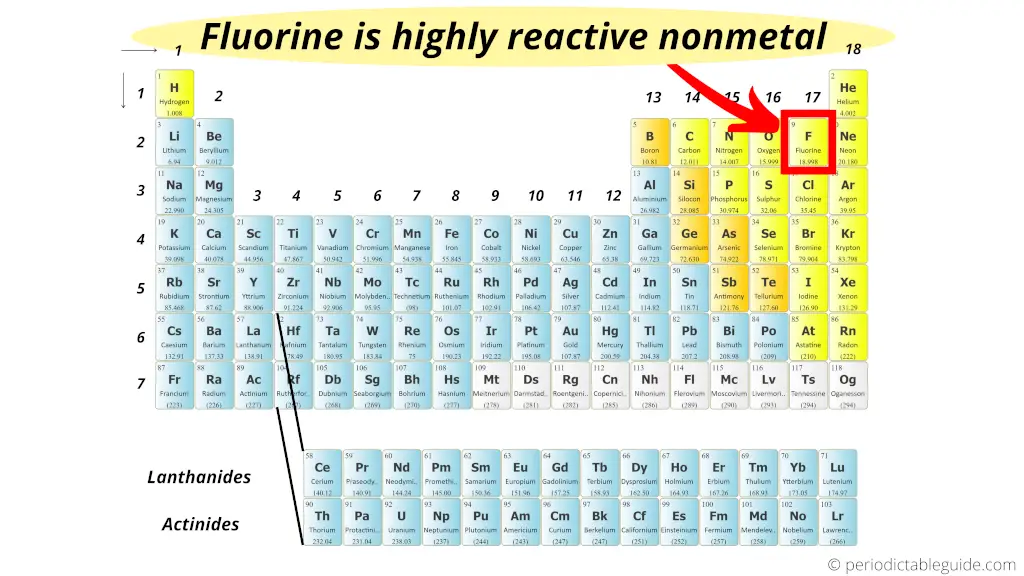
But why?
Don't worry, I'll tell you the reason behind this.
Run across, Atomic number of fluorine is 9. So it has 2 orbits and 7 electrons in its outermost orbit.
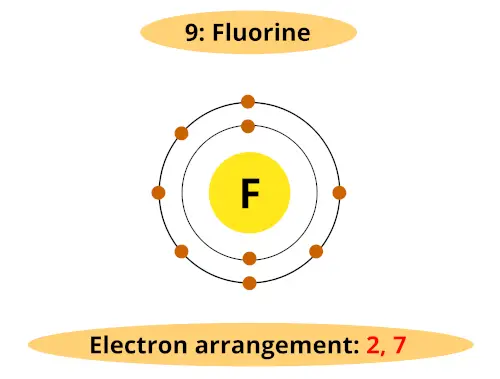
Fluorine requires only 1 electron to complete the octet. And it is very easy to gain one electron during a chemical reaction.
Also the atomic size of fluorine is very small. (Check this Periodic tabular array guide, for atomic size trend)
And because of its smaller size and demand of only one electron, information technology has the highest electronegativity.
Electronegativity is nothing but a tendency to attract the electron pair towards information technology.
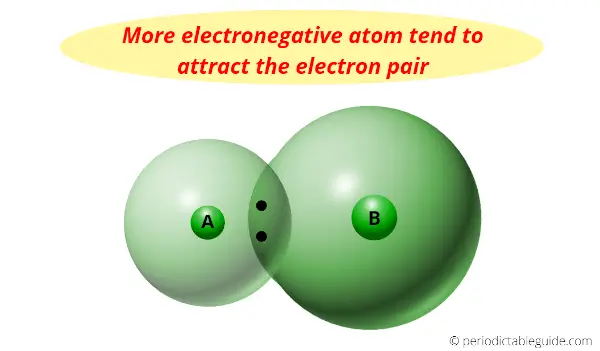
Actually the halogens are the most reactive nonmetals, but we know that equally we motility down the group, the electronegativity decreases.
In other words, Fluorine is at the height of the halogen group and information technology has less atomic size plus it needs only one electron to complete the octet.
And then it has highest electronegativity (information technology has maximum tendency to attract the electrons pair)
And because of this, fluorine has a highest tendency to react with whatsoever other element to course a compound.
Even fluorine reacts with element of group 0 like xenon, and forms compounds like XeF4 (Xenon tetrafluoride) and XeF6 (Xenon hexafluoride)
Thus fluorine is highly reactive nonmetal.
(Note: Noble gases have even smaller diminutive size compared to halogens, only they take complete octet. So they practise not participate in whatever chemical reaction. Hence noble gases are chemically inert)
Too visit:
1). Where are halogens on the periodic tabular array? (with images)
ii). Where are nonmetals on the periodic table? (with images)
Well, now I'll talk over the physical and chemical backdrop of nonmetals.
Nonmetals have exactly the reverse characteristics that of the metals.
Let's get started with concrete properties.
Physical properties of nonmetals
#1 State: solid, liquid too as gas
At room temperature, nonmetals are found in solid state, liquid land likewise as gaseous state.
- 11 non metals are in gaseous state at room temperature.
- Bromine (Br) is a nonmetal which is in liquid land at room temperature.
- Residual of the nonmetals are solids at room temperature.
#2 Little or no lusture
Nonmetals are generally non lustrous only few solid nonmetals have shiny lustrous surface (For example: Iodine have a lustrous surface)
#3 Poor conductor of rut and electricity
Nonmetals practise not allow heat and electricity to laissez passer through them.
Hence they are poor conductors of estrus and electricity.
#4 Brittle
Solid nonmetals are brittle in nature. That means they break easily when force is applied on them.
#5 Lower melting point and boiling betoken
Nonmetals have lower melting betoken also equally boiling point and thus most of the nonmetals are gaseous at room temperature.
#6 Low density
Nonmetals have lower density, hence they are calorie-free in weight.
#vii Not sonorous
Nonmetals do not bear sound. Hence they are not sonorous.
Chemic properties of nonmetals
#1 Valency
Nonmetals have four to 8 electrons in their outermost orbit. (Halogens have vii electrons and noble gases take viii electrons in outermost orbit)
#ii High electronegativity
Nonmetallic elements have the higher tendency to attract the electron pair towards it during a chemical reaction.
Hence nonmetals are highly electronegative in nature.
#iii Proceeds of electrons
Nonmetals have 4 to viii electrons in their outermost orbit. Hence nonmetals testify tendency to gain electrons during a chemical reaction to consummate the octet.
#4 Forms acidic oxides
Nonmetals react with water and form acidic oxides.
#five Good oxidizing agents
Nonmetals are oxidizing agents because they gain electron/s during a chemic reaction and get reduced.
Gratuitous Souvenir for y'all: Interactive Periodic Table
Let me tell you how this Interactive Periodic Tabular array volition help you in your studies.
1). Y'all tin effortlessly find every single detail almost the elements from this single Interactive Periodic table.
2). You will get the detailed information nigh the periodic table which volition convert a newbie into pro.
3). You volition besides get the HD images of the Periodic table (for FREE).
Checkout Interactive Periodic table and download information technology's loftier resolution image at present (It'southward Free)
Summary
Then in the entire article, nosotros have discussed the definition or verbal significant of nonmetals so we saw the position of nonmetals on the Periodic table.
Now you know that nonmetals are nowadays on the upper right corner of the Periodic table.
Then we also discussed that there are total eighteen nonmetals on the Periodic tabular array (I also mentioned a list of these 18 elements)
So we discussed about the most reactive nonmetal on the Periodic tabular array (i.e Fluorine).
And finally we discussed the physical and chemical properties of nonmetals.
I hope this article "Where are Nonmetals located on the Periodic Table" has helped yous in solving your doubts.
Let me know in the comments below, has this article helped you or not?
Suggested Important articles for you:
- Periodic table (with everything you need to know)
- Metals on the periodic table
- Metalloids on periodic table
- Halogens on periodic table
- Brine metals on periodic table
- Alkaline earth metals on periodic table
- Noble gases on periodic table
- Transition metals on Periodic table
- Inner transition metals on periodic tabular array
- What practise elements in the same grouping have in common
childresspriagenction.blogspot.com
Source: https://periodictableguide.com/nonmetals-located-on-the-periodic-table/
0 Response to "What is the only non-metal to be found on the left side of the periodic table?"
Post a Comment